Observation of skull specimen and image analysis The model-based predictions were in good agreement with experimental results, suggesting that the model captures the essential features of the mechanism of skull suture interdigitation. We numerically tested the behavior of the model concentrating on the basal effect of substrate molecules ( a 0) and verified the model with various experimental methods. Then, according to the data we defined two factors, tissue differentiation state ( u) and substrate concentration ( v), to describe the situation and formulated a simple two-species reaction–diffusion model. 
Next, we listed the molecules involved in the developmental process, and classified them into three categories depending on localization and function. First, we used human and mouse skull specimens to observe the process of suture interdigitation. In the present study, we established a simple model that can generate the interdigitated structure based on experimental data, and experimentally verify the model. Moreover, this model fails to incorporate known molecular interactions occurring in the developing suture, making it difficult to integrate the proposed fractal dynamics from the model with experimental evidence obtained from molecular developmental biology studies. However, the model assumes that the ragged edges of bones are formed before the two edges come close, which is very different from the actual dynamics ( Fig. 
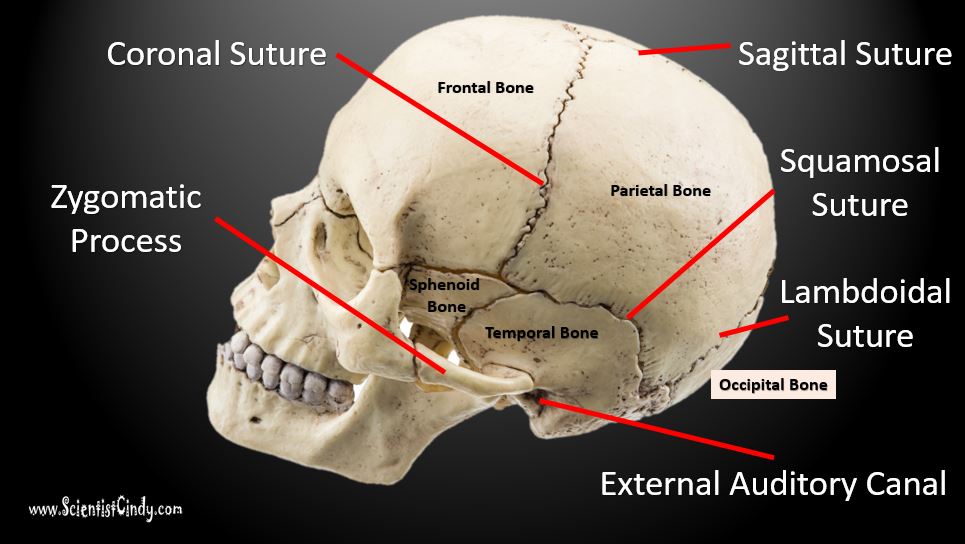
The model utilizes the Eden model, random growth of the interface, to model the formation of the interdigitated structure (Supplementary data 1). 2004 ), and a model has been proposed to explain the mechanism by which the fractal structure of sutural tissue is generated ( Oota et al. It has been proposed that, functionally, the interdigitation is related to mechanical tension ( Moss, 1961 Jaslow, 1990 Byron et al. 2008), yet identification of the molecules involved and their interactions has not led to an explanation of the mechanism of suture interdigitation. Many growth and transcription factors are known to be involved in sutural growth ( Morriss-Kay & Wilkie, 2005 Rice, 2005 Slater et al. (b) Governing equations of the system and explanations of each term. Mesenchymal cells produce substrate molecules which promote the differentiation of undifferentiated mesenchyme. Mesenchyme ( u = −1) and bone ( u = 1) are two stable states and do not interchange easily. We included two factors, osteogenic differentiation state of a tissue ( u) and substrate concentration ( v). In the late stage of growth, a complex interdigitated structure that has a noninteger fractal dimension is sometimes formed ( Long, 1985 Masuda & Yohro, 1987 Saito et al. It is not until after birth that the linear suture line begins to form a wave pattern ( Fig. Each cranial suture is first formed as a thin strip of undifferentiated tissue between two skull bones. A denticulate pattern also exists, in which small toothlike projections of the articulating bones widen towards their free ends this is seen in the lambdoid sutures ( Sperber 2001). The coronal and sagittal sutures are serrated, with the bone edges having a notched or sawlike appearance. Interdigitations found within the sutures of the cranial vault are of two types. Six primary sutures of the cranial vault exist, including the paired coronal sutures (between the frontal and parietal bones), the paired lambdoid sutures (between the parietal and interparietal bones), the single sagittal suture (between the parietal bones), and the single human metopic or murine posterior frontal suture (between the paired frontal bones). 
The mammalian skull vault consists of five principal bones (the paired frontals and parietals and unpaired interparietal), and adjacent margins of the membranous skull vault bones form the cranial sutures, in which growth of the skull vault occurs. Predictions from the model are in good agreement with experimental observations, indicating that the model captures the essential nature of the interdigitation process. This model reproduces the maintenance of thin sutural tissue at early stages, the later modification of the straight suture to form osseous interdigitations, and the formation of fractal structures. Here we show that by classifying the molecules involved into two groups, stabilizing factors and substrate molecules, complex molecular networks can be modeled by a simple two-species reaction–diffusion model that recapitulates all the known behavior of suture pattern formation. The nature of the relationship between the molecular interactions and suture pattern formation is not understood. During periods of brain growth the sutures remain thin and straight, later developing complex fractal interdigitations that provide interlocking strength. Skull sutures serve as growth centers whose function involves multiple molecular pathways. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
